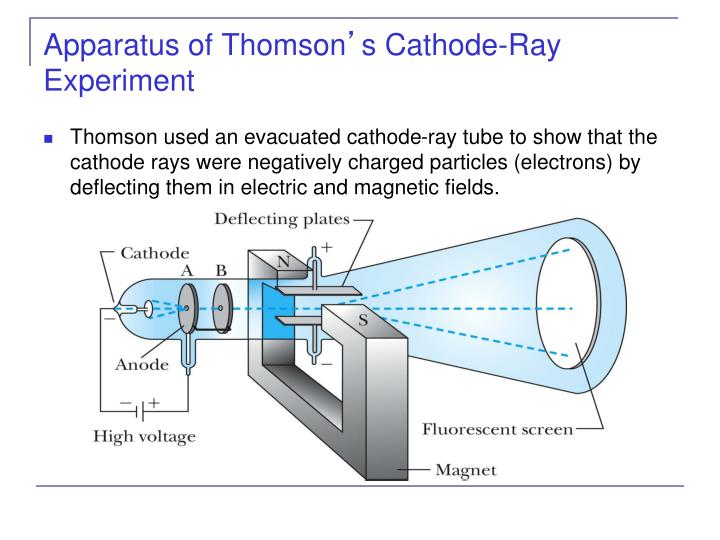
This exploration also reveals the basic inadequacy of the analytic paradigm to illuminate the conduct of science, thereby permitting many of his problems to be dissolved rather than solved. He also determined that the charge to mass ratio. Thomson during cathode ray tube experiment. 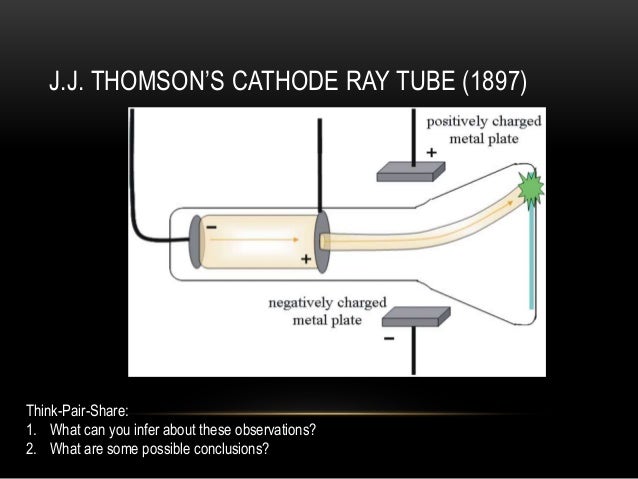
a negatively charged particle was discovered by J.J. For his work on the discovery of electrons he was awarded the Nobel Prize in Physics in 1906. Thomsons cathode ray tube experiments provided the first evidence that atoms were composed. Clarify math tasks Figure out math equation Do mathematic tasks Chemistry Unit 2: Test Flashcards In 1897, J.J. The present paper aims to use paradigmatic responses to Galison’s problems to explore the differing natures, merits and limitations of these two paradigms. Thomsons experiment revealed that cathode rays are composed of negatively charged particles called electrons. Thomson was a British physicist, and was born in Cheetham Hill, a suburb of Manchester, on 18 December 1856. This Jj thomsons cathode ray tube experiment showed which of the following helps to fast and easily solve any math problems. The paper argued for recognising at least two paradigms, one based on logic, and analytic forms more generally, the other based on deliberative judgement making. In a recent paper Hooker (Perspect Sci 26(2): 266–291, 2018b) proposed that the discipline(s) of HPS should themselves also be understood to employ paradigms in HPS to understand science, analogously to those employed in science to understand scientific domains. irst, in a variation of an 1895 experiment by Jean Perrin, Thomson built a cathode ray tube ending in a pair of metal cylinders with a slit in them. Recent research provides a basic resolution of these issues. First, he used a magnet and electrometer to observe that the cathode rays were indeed electrically charged. It is however unclear to what extent these problems, and constraints on their solutions, are of HPS’s own making. Thomson performed three experiments with cathode ray tubes. Before his work, atom was considered to be. These particles were later called electrons. Thompsons cathode ray tube experiment electricity was discharged in a cathode ray tube with electricity flowing from the cathode to the anode. 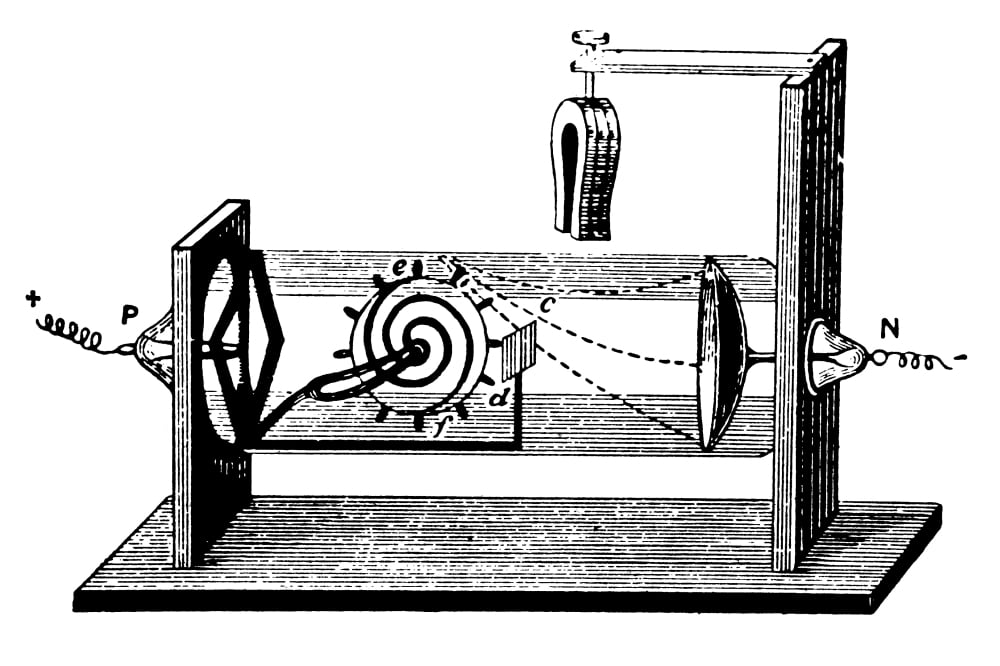
On April 30, 1897, he startled the scientific world by announcing that the particles composing cathode rays were much smaller than atoms. In an Isis 2008 review of research in History and Philosophy of Science (HPS), Galison opened discussion on ten on-going HPS problems. Thompson performed a brilliant experiment which proved that atom consisted of charged subatomic particle(s). During 1896 Thomson also conducted an investigation of cathode rays.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
